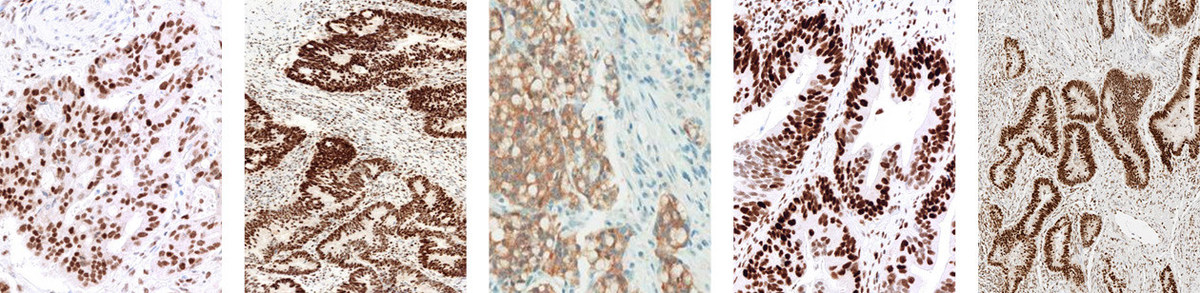
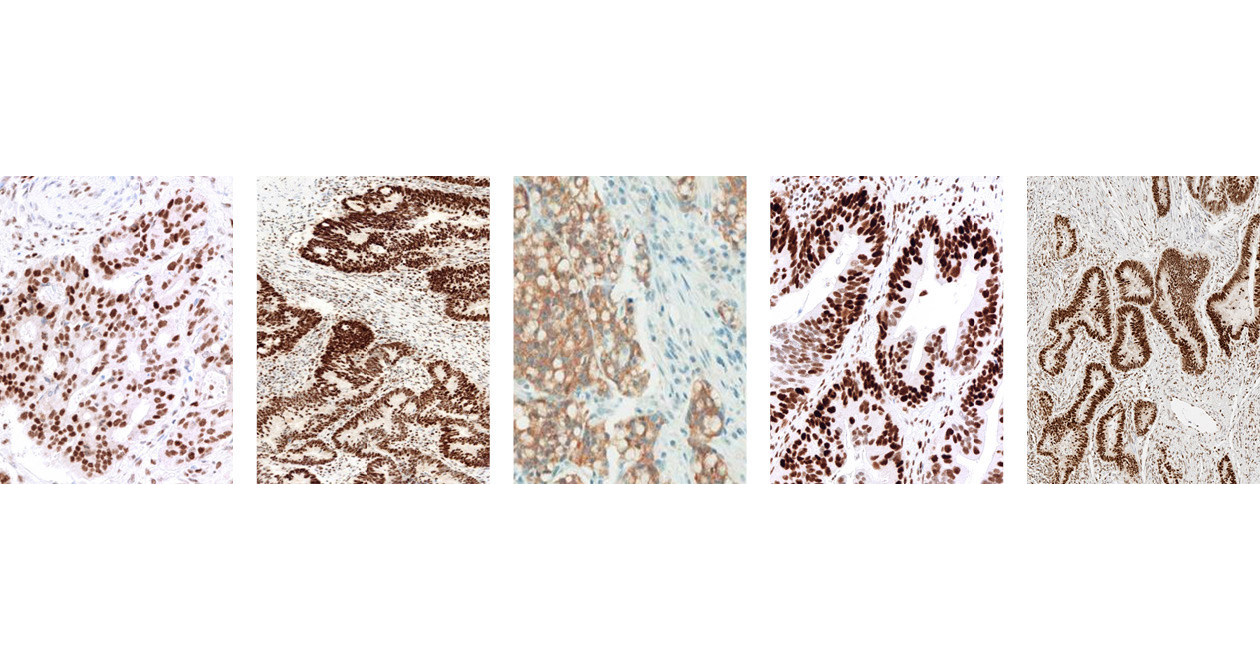
Roche Tissue Diagnostics on Twitter: "Media: VENTANA MMR IHC Panel helps identify probable Lynch syndrome, a hereditary form of colorectal cancer https://t.co/g3SZAy8Dyf https://t.co/HU2ANmTzTu" / Twitter
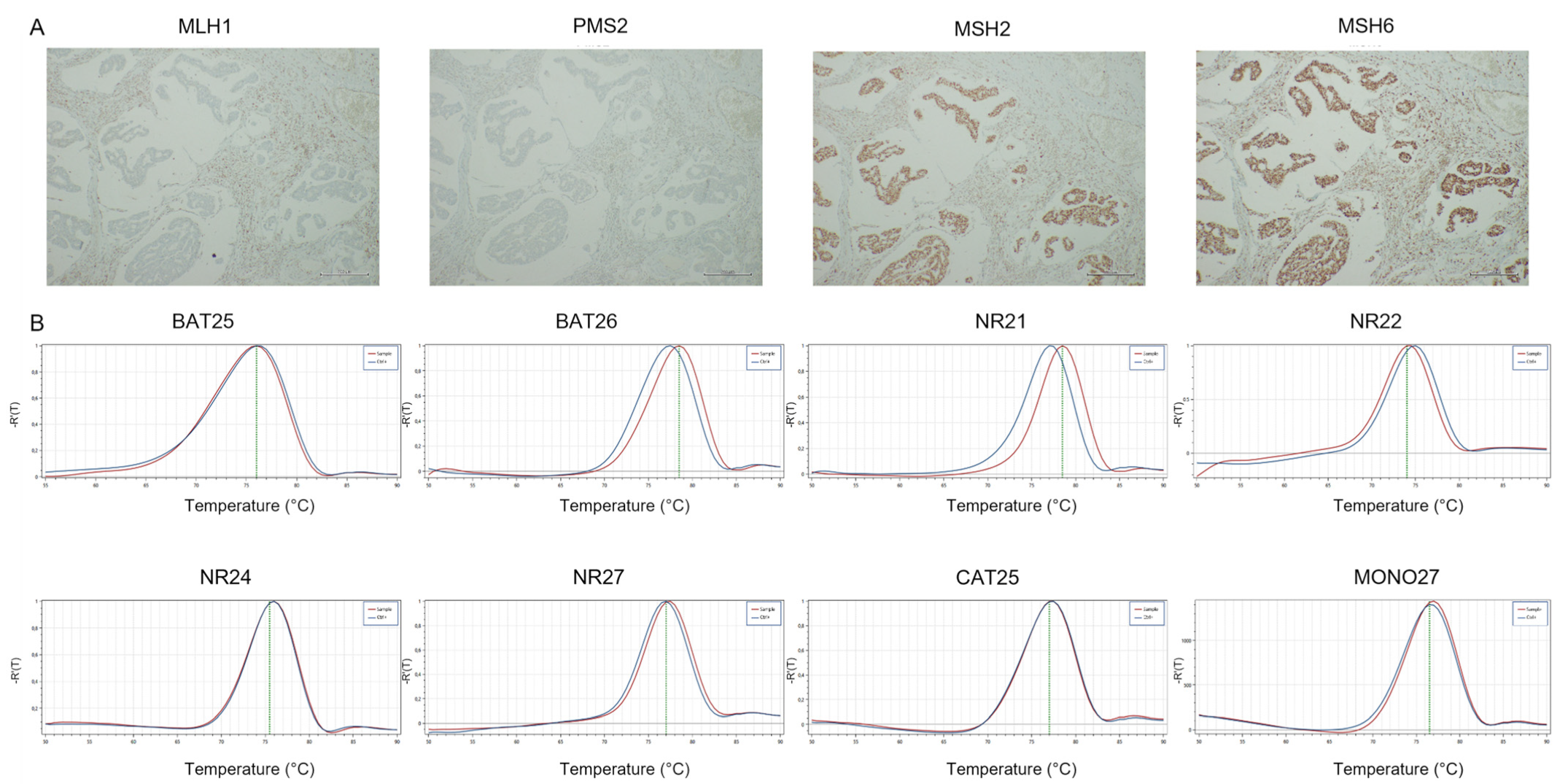
Ventana MMR IHC Panel DECISION SUMMARY A. De Novo Number: DEN170030 B. Purpose for Submission: De novo request for evaluation of

Simple IHC reveals complex MMR alternations than PCR assays: Validation by LCM and next‐generation sequencing - Amemiya - 2022 - Cancer Medicine - Wiley Online Library
Ventana MMR IHC Panel DECISION SUMMARY A. De Novo Number: DEN170030 B. Purpose for Submission: De novo request for evaluation of
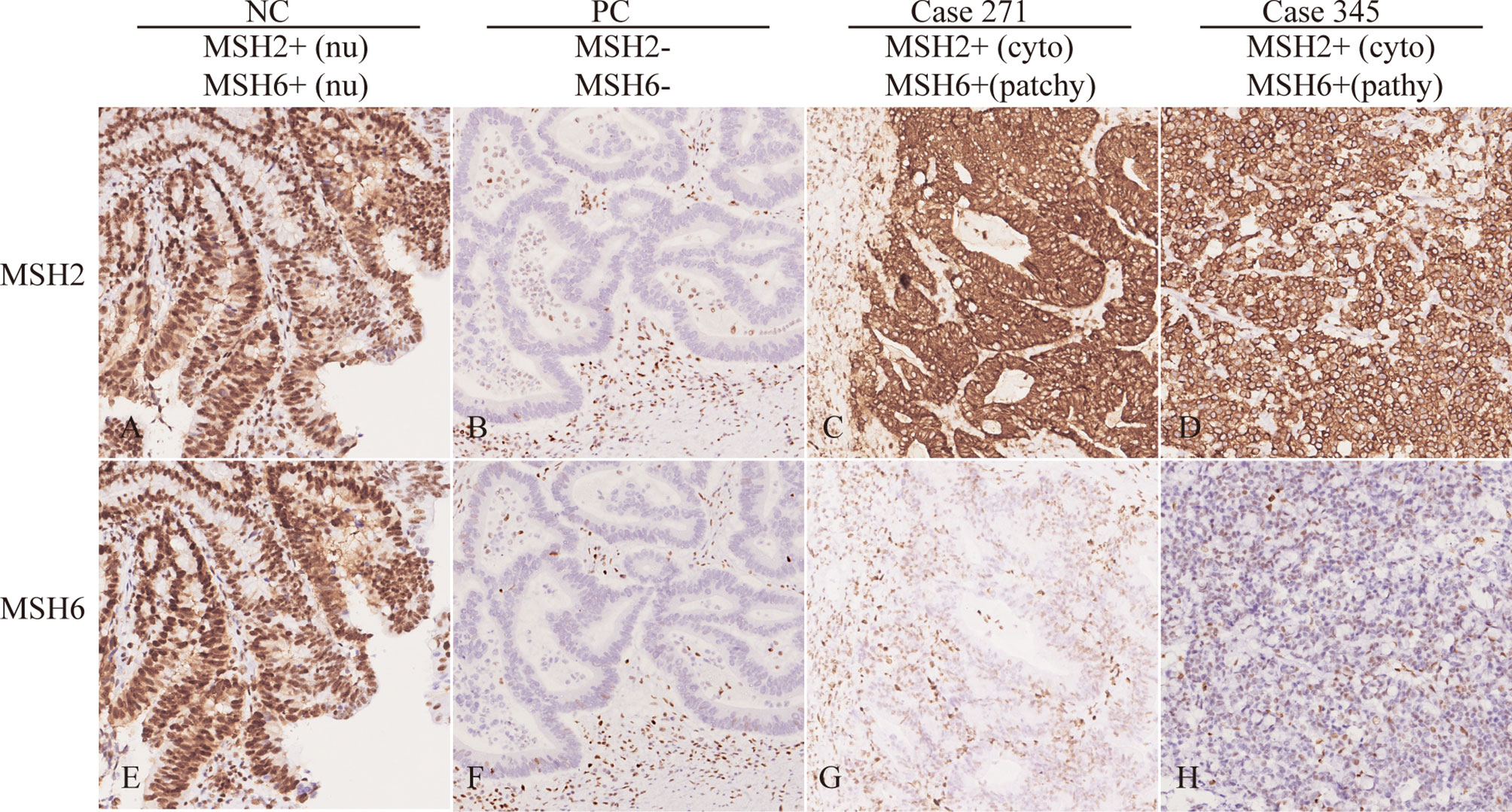
Frontiers | Cytoplasmic MSH2 Related to Genomic Deletions in the MSH2/EPCAM Genes in Colorectal Cancer Patients With Suspected Lynch Syndrome

Detecting mismatch repair deficiency in solid neoplasms: immunohistochemistry, microsatellite instability, or both? | Modern Pathology
Ventana MMR IHC Panel DECISION SUMMARY A. De Novo Number: DEN170030 B. Purpose for Submission: De novo request for evaluation of
Ventana MMR IHC Panel DECISION SUMMARY A. De Novo Number: DEN170030 B. Purpose for Submission: De novo request for evaluation of